Chemical Properties & Facts:
Nitric acid is a strong monobasic acid and an oxidizing agent, which is characterized by a colorless, yellow, or red liquid.
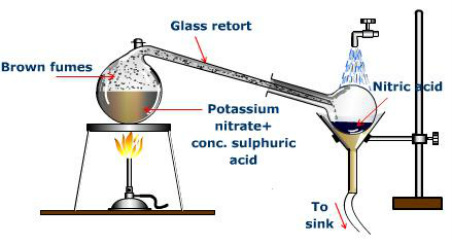
Nearly 250 years after its discovery, Wilhelm Ostwald developed the "Ostwald Process" (in 1901), which became the primary method of producing nitric acid. Ammonia gas is continuously oxidized into nitric oxide and nitrogen dioxide by air or oxygen by using a platinum gauze catalyst.
A key characteristic of nitric acid is that it creates a choking odor that is acrid and suffocating. Nitric acid is an extremely caustic and corrosive liquid capable of corrroding some plastics and rubbers. Though not flammable, nitric acid can emit poisionous oxides of nitrogen and acid fumes when heated in fire.
Nearly 250 years after its discovery, Wilhelm Ostwald developed the "Ostwald Process" (in 1901), which became the primary method of producing nitric acid. Ammonia gas is continuously oxidized into nitric oxide and nitrogen dioxide by air or oxygen by using a platinum gauze catalyst.
A key characteristic of nitric acid is that it creates a choking odor that is acrid and suffocating. Nitric acid is an extremely caustic and corrosive liquid capable of corrroding some plastics and rubbers. Though not flammable, nitric acid can emit poisionous oxides of nitrogen and acid fumes when heated in fire.
